Alzheimer’s Disease
Beta Amyloid
Alzheimer’s disease is characterized by the beta amyloid plaques and tau neurofibrillary tangles in patient brains. Genetic information unequivocally points to an important role of beta amyloid in causing damage to neurons and synapses, followed by multiple downstream cellular reactions such as neuroinflammation and neurofibrillary tangle formation.
By using PET imaging of the brain beta amyloid plaques are detectable up to twenty years prior to the development of the clinical signs of Alzheimer’s disease including memory loss, difficulties of spatial orientation and word finding, deficits in performing normal activities of daily living and problems driving a car.
Neurimmune discovered aducanumab, a human monoclonal antibody to deplete brain amyloid of patients with Alzheimer’s disease. Neurimmune originally licensed its antibody aducanumab for the treatment and prevention of Alzheimer’s disease to Biogen in 2007 and regained global rights to the program in 2024.
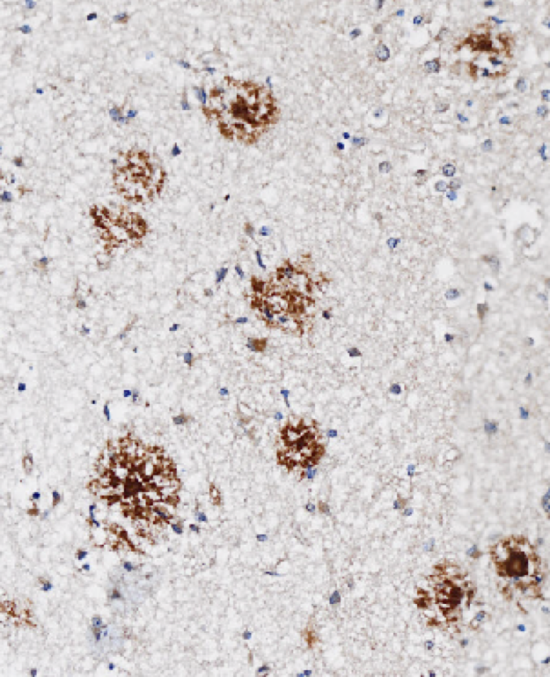
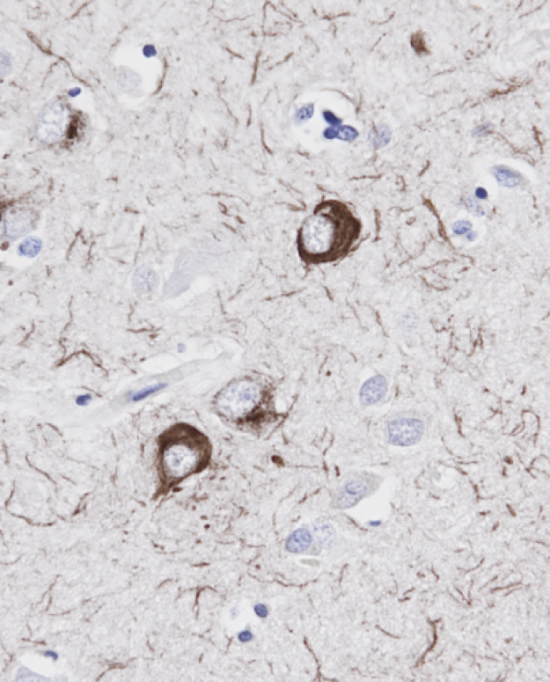
Tau
Neurofibrillary tangles in brains of patients with Alzheimer’s disease are composed of aggregated tau proteins which accumulate in nerve cell bodies as well as outside of nerve cells in the form of neuropil tau threads. Researchers believe that tau pathology can be initiated by beta amyloid toxicity but once formed can cause downstream damage to neurons independently of beta amyloid. Therefore, tau is increasingly recognized as a prime therapeutic target for treatments designed to remove tau in brains of patients with Alzheimer’s disease.

Quote
« We are entirely devoted to prevent, treat and hopefully eradicate Alzheimer's disease.»
Acetylcholine
The disease process in Alzheimer’s disease is complex. In addition to beta amyloid, tau neurofibrillary tangles, synaptic damage and neuroinflammation, cholinergic neurons connecting the basal forebrain with multiple other brain regions including the hippocampus and the brain cortex degenerate early on in the pathological cascade of Alzheimer’s disease, leading to memory loss, cognitive deficits and behavioral symptoms. The so-called “cholinergic deficit” hypothesis has led to the first approved drugs for the symptomatic treatments of Alzheimer’s disease back in the late 90ies of the last century. Neurimmune’s collaboration partner NSC Therapeutics is developing a small chemical analog of the cholinergic neurotransmitter acetylcholine (NSC001) for the next generation treatment of patients with cholinergic deficits. In addition to Alzheimer’s disease, patients with Lewy body dementia or Parkinson’s disease dementia suffer from such cholinergic deficits.
Neuroinflammation
To address the pathogenic effects of neuroinflammation in the brains of patients with Alzheimer’s disease, Neurimmune’s research is targeting several inflammatory cellular pathways to develop antibody-based treatments designed to mitigate neuroinflammation.


